Coronavirus (COVID-19) Update: FDA Takes Multiple Actions to Expand Use of Pfizer-BioNTech COVID-19 Vaccine | FDA
OCHD ANNOUNCES MODERNA AND J&J BOOSTER AVAILABLE AT VACCINATION CLINICS FOR ELIGIBLE RESIDENTS – Ocean County Health Department

Covid-19 Booster Vaccinations for Eligible Patients - Clinics October/November 2022 - Sirona Medical - Cabra, Dublin 7 - GP Practice

COVID-19 vaccination – Social – Booster doses (Aboriginal and Torres Strait Islander people) | Australian Government Department of Health and Aged Care

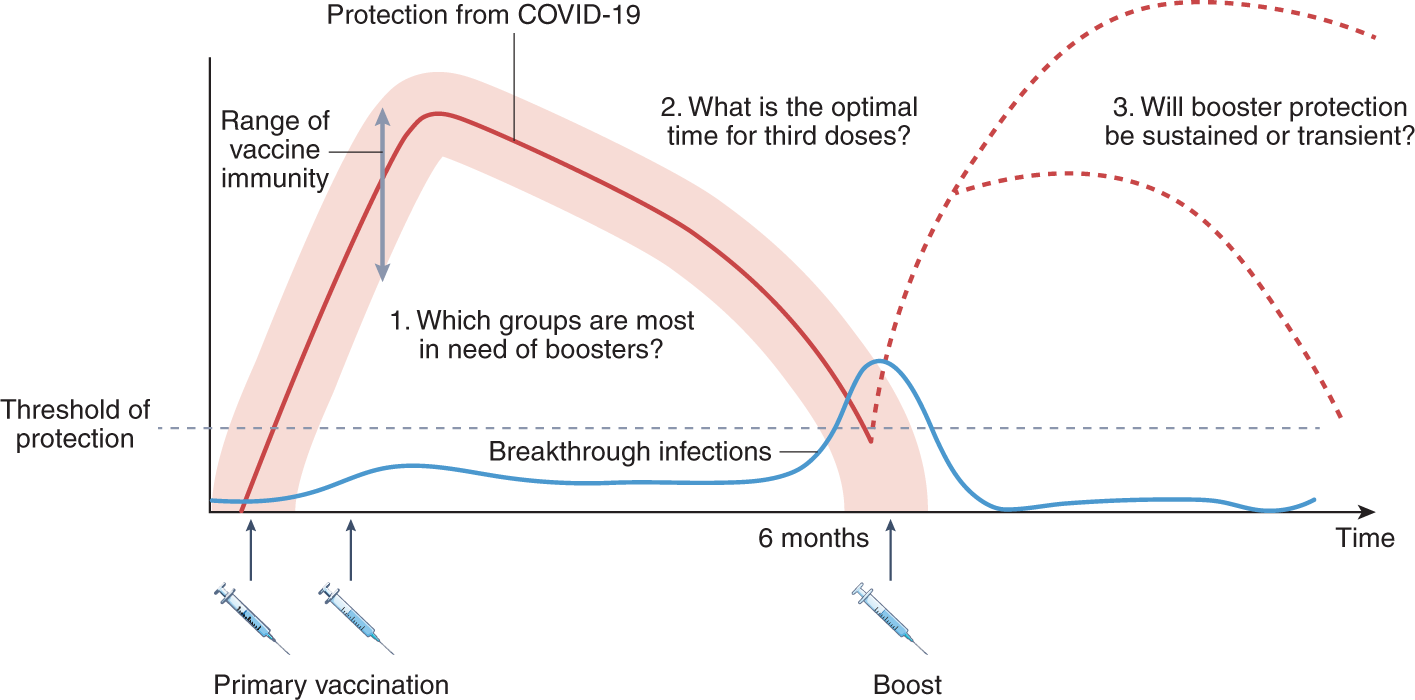
COVID-19 Incidence and Mortality Among Unvaccinated and Vaccinated Persons Aged ≥12 Years by Receipt of Bivalent Booster Doses and Time Since Vaccination — 24 U.S. Jurisdictions, October 3, 2021–December 24, 2022 | MMWR

CDC on X: "CDC strengthens recommendations and expands eligibility for #COVID19 vaccine booster shots. Read full statement: https://t.co/gGNakf1VB7 https://t.co/PBxSuZBFlx" / X

Coronavirus (COVID-19) Update: FDA Shortens Interval for Booster Dose of Moderna COVID-19 Vaccine to Five Months | FDA

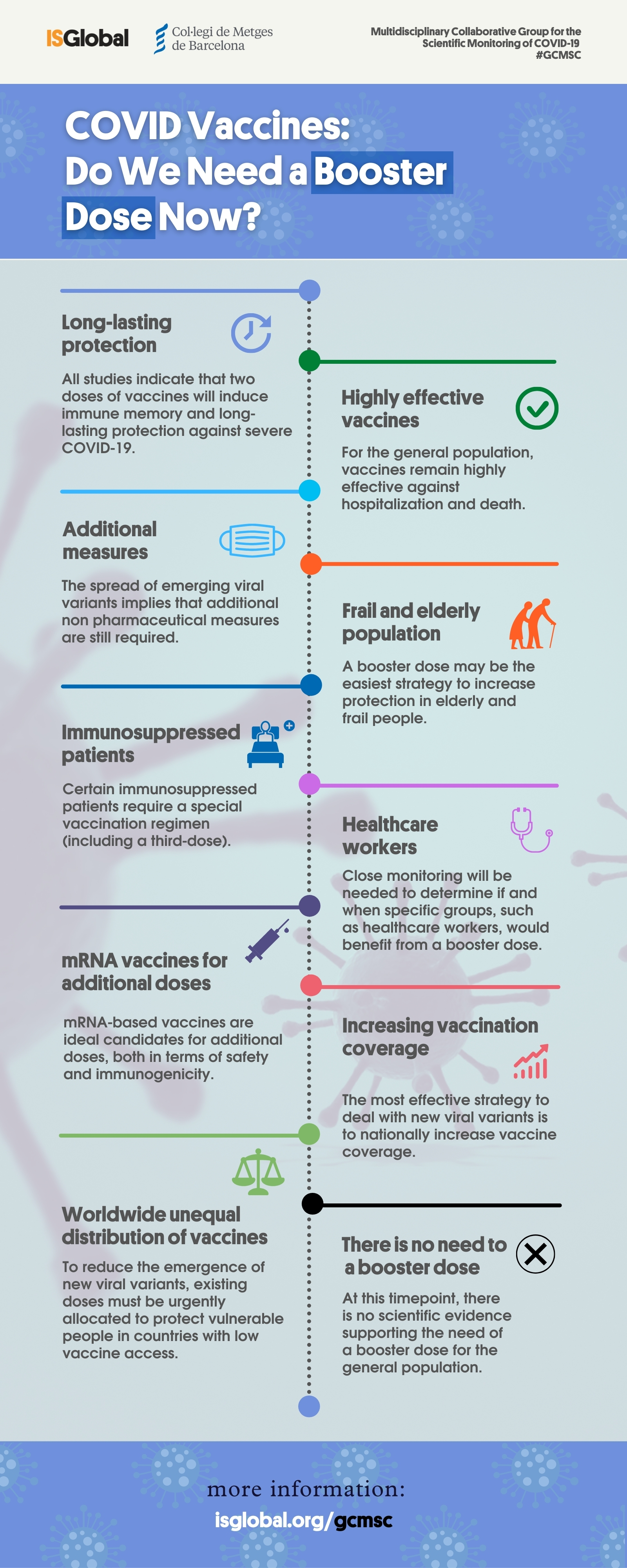
Early Estimates of Bivalent mRNA Booster Dose Vaccine Effectiveness in Preventing Symptomatic SARS-CoV-2 Infection Attributable to Omicron BA.5– and XBB/XBB.1.5–Related Sublineages Among Immunocompetent Adults — Increasing Community Access to Testing ...